The Aperio Blog
Let’s Make It Clear-Part 1: The Sponsor-CRO Relationship
LET’S MAKE IT CLEAR:
A Series About Strengthening the Sponsor-CRO Relationship

Cari Kniola
Senior Director, Corporate Strategy
April 12, 2022
CROs often get a bad rap. There’s no way to sugar-coat it, and the reputation is one that has been garnered over years of Sponsor-CRO relationships gone sour. How does this happen? Regardless of intentions and collaboration at the start of a trial, there is a point in some partnerships where they hit a catastrophic impasse. In April 2020 the Avoca Group released their 2019 State of the Industry Report on Clinical Outsourcing Spend and Key Relationship Measures that gave insights into what can cause a number of those relationship breakdowns. Some examples include:
- Change orders
- High turnover
- Inability to meet timelines/deliverables
- Quality of Oversight
- Communication
In general, The Avoca Group found four key relationship indicators: Quality, Value, Relationship, and Overall Work. Much like a marriage, when key needs of a relationship are met, the union is strong. But let one of these falter, and cracks begin to show in the foundation – and if not addressed they can cause it all to come tumbling down.
As with any relationship, the view is often skewed when it comes to relationship satisfaction. The Sponsor-CRO relationship is no exception, as demonstrated by the graphic below from the same Avoca Group report.
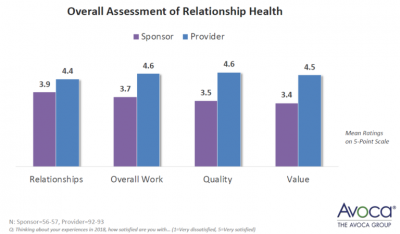
What could be the cause of such a varied perception of the success of the Sponsor-CRO relationship? In conversations had with sponsors who are contemplating leaving their current providers, a common thread is ‘I thought x, but they did y’. In other words, they weren’t on the same page. That page could be from any chapter out of the clinical trial execution handbook – units in a budget, who will be on the study team, what leads to a change order, who is responsible for what tasks, communication breakdowns. Any one of these alone or in combination with other misunderstandings will start to fray trust and confidence, not just in the services being provided, but in the integrity of the relationship.
We hate to hear these stories. We hate it for the sponsors who are frustrated and disappointed and now proceeding with extreme caution, and we hate it for CROs because it makes all of our jobs harder. So how can we fix it? How can we start to get on the same page? With the CRO market expected to have a CAGR of 12.1% over the next 7 years, these relationships aren’t going anywhere. So how can we make them better?
That is the goal of our new blog series, Let’s Make it Clear – to provide information that helps strengthen the Sponsor-CRO relationship. Within this series, we’ll address some common sponsor pain points, discuss how these often come about, and talk about ways they can be avoided and / or worked through. Our goal with this series is to provide insights that will help remove ambiguity and start everyone in the Sponsor-CRO relationship off on the same page, regardless of the CRO they use. Sure, we’d love that CRO to be us – but we aren’t a fit for everyone. And the more successes sponsors have with their CRO of choice, the easier it gets for all of us to start off the Sponsor-CRO relationship with less baggage, more trust, and a stronger foundation.
Join us in the launch of Let’s Make it Clear with our first article Not a Time for Speed Dating. We look forward to growing Sponsor-CRO relationships together!
About Aperio Clinical Outcomes
Aperio Clinical Outcomes provides full, customizable clinical research services across multiple therapeutic areas. Dedicated to transparent interactions with clients, Aperio also offers consulting services in Quality Assurance, Strategic Resourcing, and practical application of the latest Clinical Trial Technology.
In an industry flooded with data, metrics and technology, Aperio remains steadfastly focused on the most important part of the process: people. From start-up to close out and every point in between, Aperio works with its partners to deliver outcomes that will improve patient care and save lives.
Aperio Clinical Outcomes.
We make it clear.
